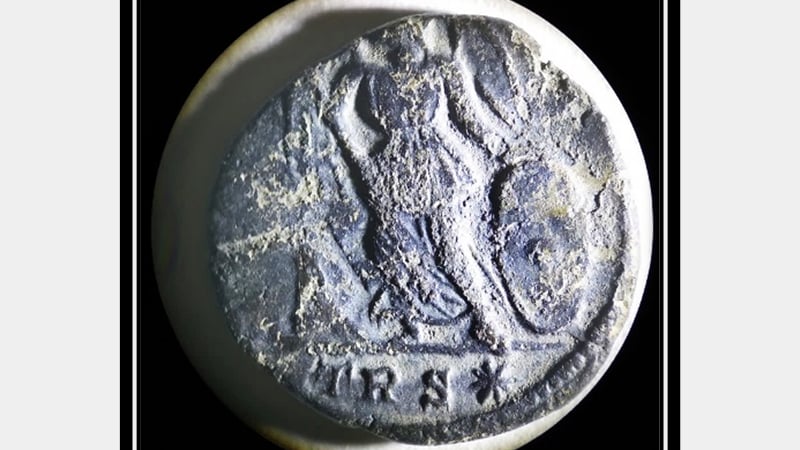
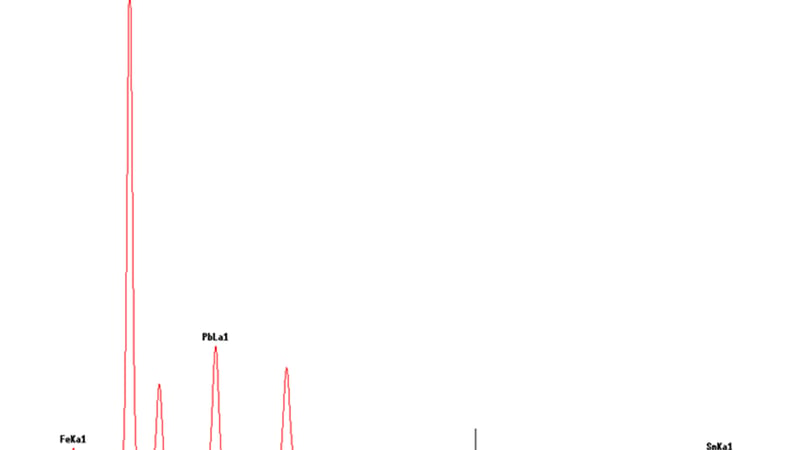
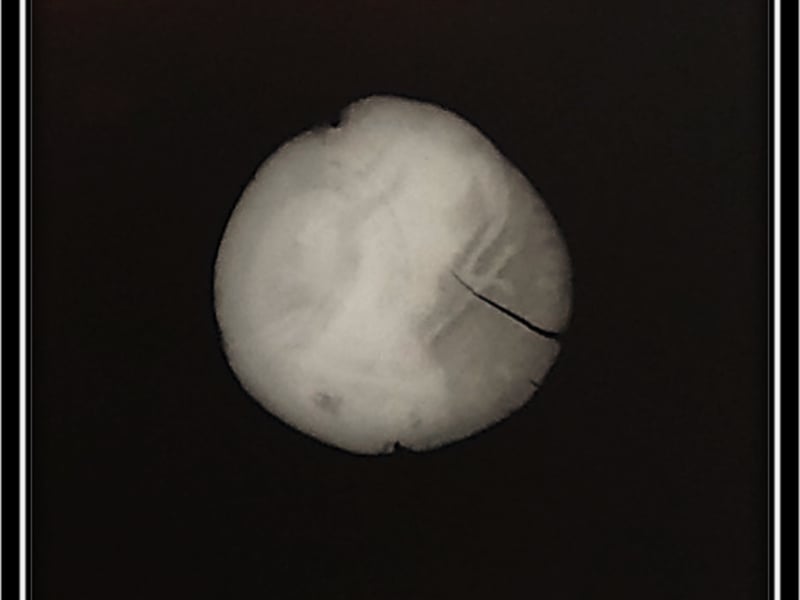
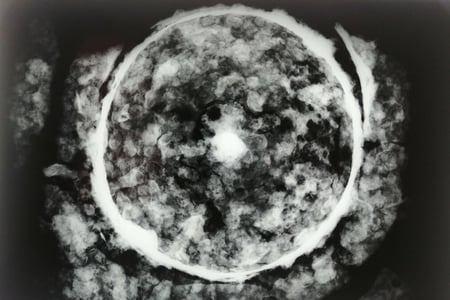
Conservation considerations: A Roman Coin
By Natasha Waddell, MA Conservation Studies student, specialising in Metals
This blog post is being used as a tool for working through my thought and decision-making process when finalising my treatment proposal for an archaeological Roman coin. This is the first time I will be working on an object of archaeological context. As incredibly excited as I am, the whole process is brand new.
I will start off by discussing the context of the coin and will then walk you through my thought processes when analysing the condition of the artefact. This will guide my final reflection on a possible treatment proposal.