Epoxy and Elephants: An Adhesive Adventure
By Kate Jennings, GD Metals
By Kate Jennings, GD Metals
One of the odd things about being a metals conservation student is that you seem to use adhesives far less than other departments. When presented with one of my recent objects to work on, my initial reaction was therefore to try to ignore that tiny voice at the back of my mind which was telling me adhesive was the only way to go. Before we get into that though, let me introduce the object in question so that you can understand a bit more about my dilemma.

Meet the "Lelephant"-so called because he is a small statue of an elephant made from almost pure lead. He belonged to the family of one of the staff who works at West Dean, and was donated to the Metals Conservation Department for a student to work on because, as you can see, he has undergone a fairly major calamity.
Most of the issues with the elephant derive from whatever incident caused his hind leg to become detached. These include (a) the detachment of the leg itself, (b) a number of cracks radiating out from the detachment point, (c) deformation of the main body and leg and (d) some loss of material around the break points.
The challenge I have been set is to find some way of reattaching the leg. There are a number of complicating factors standing in the way of completing this challenge, the most significant of which are that the wall thickness of the elephant's body is only about 0.7-0.9mm, but the leg has been filled with lead and so weighs in at a hefty 380 grams. This means there is a huge amount of structural work being done by very thin walls.
It also makes the thought of using even very soft (i.e. very low melting point) solder to carry out a hot repair to this object even less appealing than using adhesive-the risk of melting such thin section is significant.
So the decision was made-adhesive it would be.
In order to approach this task logically and also to make the most of this learning opportunity, the preparatory work was broken down into several distinct tasks as follows:
Key requirements
When working with lead objects it's important to know that as a material lead is particularly sensitive to organic acids (especially acetic acid), and also has the general vulnerabilities of all metals to high humidity and galvanic corrosion (increased corrosion rate if in electrical contact with other metals that are less reactive). This means any adhesive used should not be water based and also must not give off acetic acid as a degradation product.
The key features of the join to be created must be considered in addition to the quirks of the materials being worked with. In conservation it is often the case that joins will be designed so that these will break preferentially to new areas in the object itself-this helps minimise the chance of further damage to objects should they undergo any more accidents. In this case there is a difficult dynamic to manage of needing to make sure the join can support the (not inconsiderable) weight of the elephant's leg, but also is not so strong that if the elephant were to be dropped again the join would remain intact and the elephant would break somewhere else.
Two other key requirements that were identified during initial brainstorming were that the join will probably need to be given some form of structural support-the thinness of the walls means that if adhesive is just applied to the edges of these the join area will be a tiny butt joint that is only 0.7-0.9mm wide along its entire length. It was therefore decided that some form of bridging support must be incorporated into any join. Given the irregular morphology of the elephant's walls the ideal bridging material would be mouldable initially so that it can be fitted precisely, but then hardens fairly quickly so that it will retain these shapes without having to be held in place for long.

In summary, the key requirements identified were:
Adhesives - Back to basics
First things first - what is an adhesive?? Most dictionary definitions revolve around two key concepts when defining an adhesive - (1) it's a substance, and (2) it holds two (or more) other items together. Maybe an over-simplification, but useful to understand where you're starting from.
Starting to drill down into the detail of what that actually means, the key question is how do adhesives hold those other items together? The answer to that question is hugely complicated, with several competing and/or complementary theories (depending on your point of view) presenting different models - these include:
Mechanical interlocking.
Electronic theory.
Boundary layers and interphases.
Adsorption (thermodynamic) theory.
Diffusion theory.
Chemical bonding theory.
In reality, the types of mechanisms set out in these theories may be present in all sorts of combinations in any given adhesive bond, so they can all provide useful information about how and why adhesives work at different levels.
As this is a blog post, we will keep it simple though - the key ideas to be aware of to start understanding adhesion theory are (1) the role of adhesive and cohesive forces in adhesive bonds and (2) why it's useful to understand the concept of surface wetting.
Adhesive and Cohesive Forces
Adhesive and cohesive forces are important to understand in order to be able to recognise the levels of interactions that happen in joints held together by an adhesive. Cohesive forces are what keep like molecules together, whereas adhesive forces will keep different molecules together. If we take, for example, two pieces of wood and glue them together, cohesive forces are the internal forces keeping each of the pieces of wood and the glue (as separate objects in their own rights) together. Adhesive forces are what would make the glue stick to the wood. The diagram below sets these concepts out more visually.

Adhesive forces may be generated by different means (each of the theories mentioned above has their own ideas on this topic), but in this case they are being mentioned because they can be used to recognise and characterise what is happening in different zones of adhesive joints. This is particularly important when it comes to understanding why adhesive joints have failed, because failure may occur in adhesion zones or cohesion zones. Being able to recognise specifically where failure has occurred means the design of adhesive joins can be modified and made more effective.
Surface Wetting
Surface wetting relates to the ability of liquids to make interfaces with solids. The extent to which liquids will "wet" a surface is again determined by cohesive and adhesive forces. If the molecules within a liquid are much more attracted to each other (cohesive forces) than to the molecules within any solid surface on which they are sitting, then the liquid will tend to ball up and sit away from the solid surface. If, on the other hand, the forces of attraction between the liquid molecules and solid molecules (adhesive forces) are stronger than those within the liquid it will spread across the solid surface and "wet" it effectively. The balance between these forces can be assessed by looking at the surface tension of liquids (the state most adhesives will be in when applied) and surface energy of solids.
Surface tension of liquids effectively just refers to the surface "boundary" that will form on liquids as a result of the cohesive forces acting within a liquid and also the air around it-when there is a high surface tension, the liquid is much more attracted to itself than anything else. Such liquids will resist spreading out or penetrating thin gaps. A useful way of assessing surface tension is to look at the contact angle between a liquid and a solid surface - this concept is shown diagrammatically below.
When you talk about the surface energy of solids, you're referring to the amount of energy that it takes for the molecules that make up the main bulk of the solid to hold onto the molecules at the surface of the solid. The molecules at the surface of solids are only connected to the rest of the solid from beneath rather than from all sides (as would be the case for molecules within the main body of solid objects), so this means it takes more energy to hold onto these surface molecules. It's this energy (together with the surface tension of the chosen adhesive) that dictates how well adhesives will wet the surface.
It's worth noting that if a surface is contaminated (with an old barrier layer for example) this can change the surface energy of a solid. The physical characteristics of any surface (e.g. roughness) will also have an impact on how well adhesives will wet a surface.
In practical terms, this means that if we know the surface tension of an adhesive is lower than the surface energy of the solid to which it is applied, it will wet it effectively - key to forming a good adhesive bond.
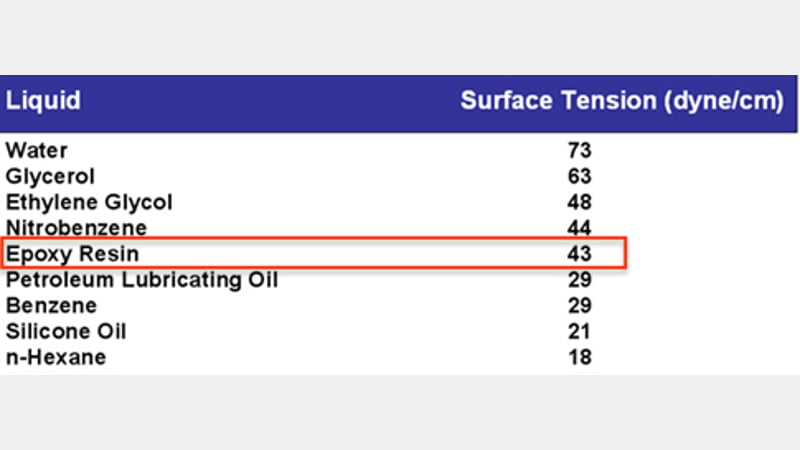
Hard solids (such as metals and ceramics) have high surface energy as a result of the types of molecular bonding (cohesive forces) present. Below are two table showing (respectively) examples of surface tensions and surface energies - this clearly shows that the surface tension of epoxy resins is significantly lower than that of lead. Epoxy resins are therefore a good choice of adhesive for a lead substrate, and will be a good starting place when selecting adhesives for the repairs to be made to the elephant.
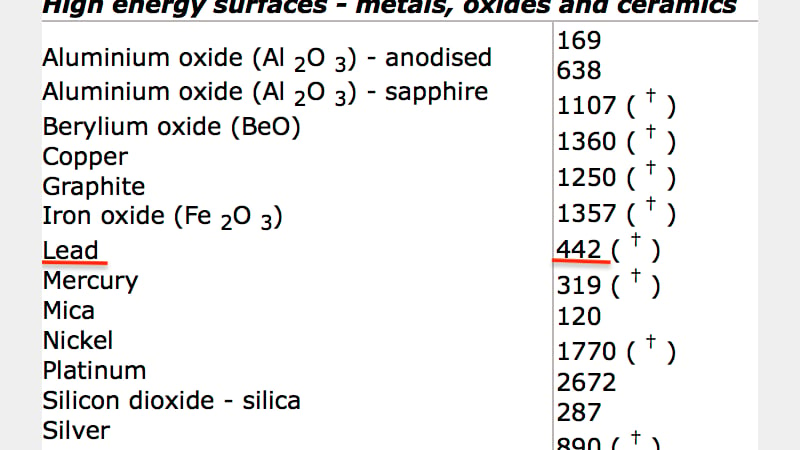
Interim Conclusions
Congratulations on making it to this point! Hopefully this blog post will have helped two different audiences in slightly different ways. For conservators, maybe a refresher (or introduction) to some useful underlying concepts relevant to many different materials and practices. For non-conservators, an illustration of the kind of level of detail conservators will go into when working on your objects, and why it's important to make sure you have someone with the right kind of experience and knowledge.
This section is titled "Interim Conclusions" because this is only the start of the process of treating this particular object. There will therefore be a second post reporting on the preliminary testing of different adhesives and bridging materials, and a third part that will (finally) outline the actual repairs made to the elephant himself. Each will be self-contained, so don't feel like you have to read all three for them to make sense - although of course you are more than welcome to! (We will add links here when the subsequent parts are written.)